Weight-Loss Drugs Suppress Tobacco Cravings
The finding may impact tobacco sales, given the projected growth for such medications, according to Morgan Stanley.
The finding may impact tobacco sales, given the projected growth for such medications, according to Morgan Stanley.

Even in its watered-down version, the recently approved EU supply chain law will impact tobacco companies.


Growth was driven in part by a revival of Indonesia’s hand-rolled kretek segment.


Health Ministers are currently debating Europe’s Beating Cancer Plan in Brussels.


In the U.S., restrictions on advertising are at the discretion of social media companies.


Among the proposed labels is a message that nicotine pouches are not a safe alternative.


G.L.J. de Best will take over from Denis Gorkun, who has been president since 2019.


The U.S. Food and Drug Administration is seeking about $20,000 from each.



Manufacturing, selling, importing and advertising has been banned since 2009.
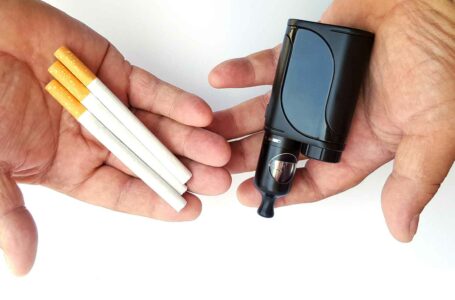

While vapes aid in cessation, more should be done to reduce their appeal to nonsmokers.


The FDA should not enforce rules declared unlawful, says civil liberties group.


Cahyadi will succeed Vassilis Gkatzelis, who will join PMI’s executive leadership team.



Tobacco Reporter’s sister publication, Vapor Voice, celebrates its 10-year anniversary.


CTP’s inability to enforce its rules often leaves state regulators and businesses baffled, writes Rich Hill.


Ling Chengxing accepted bribes and abused his authority, says anti-graft watchdog.


‘Reusable’ products won’t help the environment, say critics.


Click on a country to view its latest news from the tobacco and vapor industries.






Heated-tobacco products continue to gain momentum, although consumption patterns are shifting.


COP10 is unlikely to significantly accelerate progress toward the FCTC objectives.


Vice Ventures has carved out a niche investing in good companies in “bad” industries.


Pondering the industry’s remarkable longevity as Tobacco Reporter celebrates its 150th anniversary.


A supplier of product labeling solutions claims its technology had helped shrink the Russian illicit cigarette market by a quarter.


Three years into its five-year strategy, Imperial Brands reports progress on multiple fronts.


McKinney Specialty Labs has the expertise to generate trustworthy data and the discretion to share it only with authorized users.


Cuba’s cigar industry is on the road to recovery after several challenging years.


This year marks our publication’s 150th anniversary. To celebrate, our April 2024 issue reflects not only on a century and a half of editorial excellence, but also on the remarkable endurance of the nicotine business in an increasingly challenging operating environment. What is the secret of its success?
Unfortunately, there was less to brag about by the parties to the Framework Convention on Tobacco Control, which held their 10th meeting recently. As Stefanie Rossel reports in this issue, the Panama gathering featured lots of grandstanding but marked little progress toward the treaty’s goal of reducing the health impact of smoking.
Cheryl Olson investigated where the industry stands with ESG. Even as the term is falling out of fashion in some quarters, she found that the nicotine business remains firmly committed to the concept.
Timothy Donahue reports from the Habanos cigar festival in Cuba and Catharine Dockery shared with us the business strategy of her Vice Ventures fund, which specializes in good companies in supposedly bad industries.
Also in this issue: A profile of McKinney Specialty Labs in Richmond.



Even in its watered-down version, the recently approved EU supply chain law will impact tobacco companies.



Tobacco Reporter’s sister publication, Vapor Voice, celebrates its 10-year anniversary.



Brazilian lawmakers mull regulation of e-cigarettes, which despite being illegal remain widely available in the country.


Boosting efficiencies may help tobacco companies cope with tough times, according to CME Automation.


Banning e-liquid flavors may not achieve the outcomes that proponents of that measure have in mind.


Odiri Erewa-Meggison reflects on the significance of BAT’s Ibadan factory in Nigeria.


Though a generational tobacco ban is not without merit, the U.K. will not allow it to work in the way that it could—and thus the measure should be shelved, writes George Gay.


The company partners with 12 of the world’s leading vaping companies.


Filtrona partners with Greenbutts to expand its range of sustainable filter offerings.


The broken U.S. new tobacco product application process revealed by the numbers


Intangible assets remain a major contributor to the real value generated by the tobacco industry.


Two years into the Ukrainian conflict, businesses in the tobacco industry still scramble to adapt.