top news
-

Ohio Court to Weigh Cities’ Tobacco Ban Rules
The court’s decision is expected to clarify the scope of municipal “home rule” powers in Ohio and could influence how local governments regulate a range of industries and products.
-

Philippine Health Groups Call for Science in Nicotine Control
The growth of unauthorized vape sales emerged as a key issue, with participants calling for stronger enforcement.
-

Charlie’s Holdings Touts Progress to Shareholders
The company said it strengthened liquidity and operations over the past year, including the $7.5 million sale of 16 PMTA-authorized products.
-

Dutch Report Raises Concerns Over Big Tobacco Getting into Cannabis
Researchers cited in the report raise concerns about industry-funded studies and communications shaping cannabis narratives.
-

Pentagon’s Nicotine Policy Called into Question
The article concludes that the DOD must better align clinical policy with operational reality to maintain credibility.
-

Zyn Expanding Flavors in the Philippines
The update broadens the brand’s existing strength architecture in the market, which now spans 1.5mg, 3mg and 6mg across multiple flavors.
ATNF 2026
-

ATNF Talks Challenges in Communicating Tobacco Harm Reduction
Public health experts, physicians, and industry representatives to examine why reduced-risk messaging is failing to reach adult smokers.
-

Farrelly, Industry Experts Debate Ways to Improve PMTA Pathway
The discussion focused on the disconnect between the framework and real-world market dynamics, with panelists agreeing the current system needs to improve.
-

ATNF Panel Focuses on Harm Reduction for Adult Smokers
The discussion challenged prevailing narratives in public health, particularly the strong emphasis on eliminating all nicotine use and the skepticism toward dual use.
-

FDA, Industry Leaders Explore Product Standards at ATNF
The discussion centered on whether clearer product standards could reduce uncertainty in the PMTA process, shorten timelines, and create more predictable pathways.
-

ATNF Explores Shifting Dialogue in Tobacco Harm Reduction
The panel focused on the evolving relationship between public health and the industry, noting a gradual shift from outright exclusion toward cautious engagement.
-
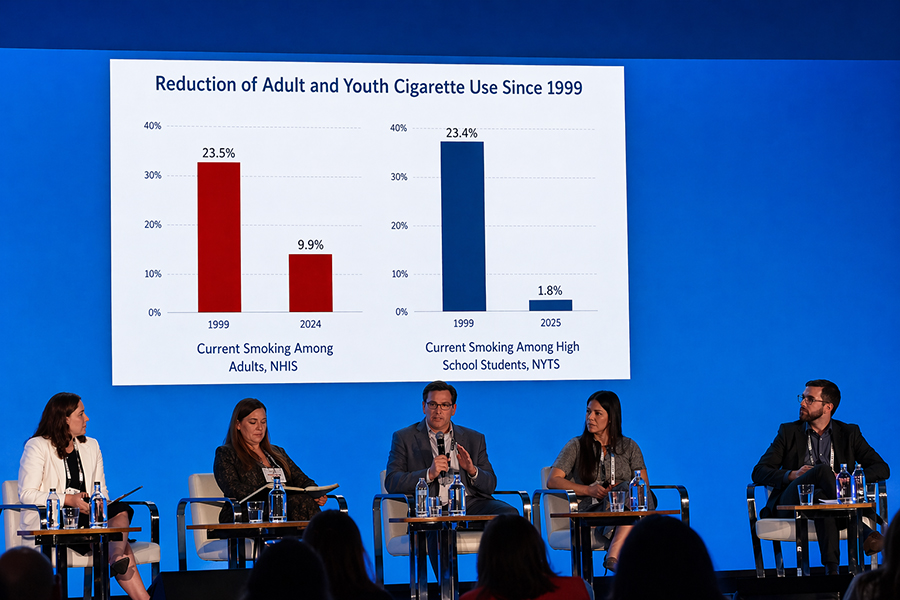
ATNF Panel Examines State-Level Policy Challenges for Harm Reduction
Speakers noted that policymakers are often making decisions without a clear grasp of existing frameworks, definitions, or scientific standards.
Global Regulation
-

Oregon Expands Tobacco Definition to Curb Youth Nicotine Access
Starting tomorrow (June 5), Oregon is expanding what counts as a tobacco product under state law, KPTV reports. The change means oral nicotine pouches, nicotine gum, lozenges, and other nicotine…
-

New York Introduces Bill for Generational Nicotine Ban
New York State has formally introduced a generational nicotine ban that would make it illegal for any person born after December 31, 2007, to purchase nicotine products, including non-combustibles, in…
-

Senators Press Altria, Reynolds Over Lobbying
Six U.S. senators, including Democratic whip Dick Durbin and Elizabeth Warren, sent public letters to Reynolds American and Altria asking questions about donations to and lobbying of the Trump administration,…
-

New York Taxes Nicotine Pouches at 75% Under Enacted Budget
Nicotine pouches in New York will now be taxed like other tobacco products such as cigars, following Governor Kathy Hochul’s signing of the state’s $268 billion budget into law on…
-

Brazil Readies Crackdown on Crime in Betting, Tobacco Sectors
Brazil’s government is in the final stages of preparing operations targeting organized crime in the online betting and tobacco sectors, a senior government official with direct knowledge of the matter…
-

Former HHS Secretary Urges FDA to Reach the “Forgotten Smoker”
In an opinion piece, Dr. Tom Price — a physician, former member of Congress, and former Secretary of Health and Human Services who spoke at ATNF 2025 — argues that…
Agriculture & Sustainability
-

Pakistani Tobacco Traders Threaten Company Blocks
Traders are demanding restoration of more than 5,000 grower contracts, reinstatement of last year’s procurement quota, and greater intervention by the Pakistan Tobacco Board.
-

Malawi Tobacco Commission Faults Farmers for Poor Handling, Rejections
Malawi’s Tobacco Commission has issued a stern warning to tobacco farmers over poor handling practices that it says are damaging the country’s leaf quality, depressing prices, and driving up rejection…
-

Malawi Tobacco Farmers Facing 91% Rejection Rate
Farmers claim the system unfairly favors contract tobacco, while independent growers face repeated rejections and mounting transport costs.
-

Zimbabwe Reminds Tobacco Farmers Stalk-Destruction Deadline Passed
TIMB is also urging farmers to conduct soil testing before planting to optimize fertilizer use, improve leaf quality, and reduce input costs.
-

CORESTA Extends Abstract Deadline
The event is calling for papers in Agronomy & Leaf Integrity, Phytopathology & Genetics, Product Science, and Product Technology.
-

Zimbabwe Tobacco Eyes Record Output, Concerned About China
Growers report increasing concerns over debt exposure, fees, and pricing, particularly within systems dominated by Chinese contractors.
Science & Innovation
-

One in 10 vape users still have stockpiled disposables
The UK ban sparked a massive stockpiling effort, with up to 70% of respondents admitting they bought and stored disposable vapes.
-

Study: Collective Trauma Reminders Spark Tobacco, Cannabis Cravings
New research from the Hebrew University of Jerusalem and the Israel Center for Addiction and Mental Health finds that merely being reminded of a collective trauma can trigger an immediate,…
-

Study Finds Herbal Cigarettes May Match or Exceed Tobacco’s Harms
A new study from researchers at IIT Bombay and the University of Illinois Urbana-Champaign challenges the assumption that “tobacco-free” and “nicotine-free” cigarettes are safer to smoke, finding that herbal cigarettes…
-
UK Price Cap Could Raise Billions and Cut Deaths, U. of Bath Study Finds
Research published by the University of Bath Tobacco Control Research Group and the University of Sheffield’s Addictions Research Group provides the first real-world modelling of a proposed “polluter pays” tobacco…
-
Graphic Cigarette Warnings May Nudge Smokers Toward Vaping, Study Finds
New research from Washington State University, published in the Journal of Business Ethics, finds that graphic anti-smoking warnings on cigarette packaging may produce an unintended consequence: rather than quitting, some…
-

Japan Review Raises Questions Over HTP Risks
A government panel claimed that some devices may produce higher levels of certain carcinogenic substances than conventional cigarettes.

Business & Finance
-

Charlie’s Holdings Touts Progress to Shareholders
The company said it strengthened liquidity and operations over the past year, including the $7.5 million sale of 16 PMTA-authorized products.
-

Zyn Expanding Flavors in the Philippines
The update broadens the brand’s existing strength architecture in the market, which now spans 1.5mg, 3mg and 6mg across multiple flavors.
-

KT&G Continues Growth with International Investors
The company’s appeal is being driven by both capital returns and strong operating performance.
-

New Article Highlights Premium Cigar Shift
The piece argues that cigars are increasingly aligned with modern consumer preferences for discretion, expertise, and immersive hospitality environments.
-

Pyxus Reports Record Adjusted EBITDA, Strong Q4
Pyxus International reported results for its fourth quarter and fiscal year ended March 31, 2026, highlighting a 35.2% year-over-year increase in fourth-quarter net sales, full-year operating income of $162.7 million…
Around the Industry
-

Ohio Court to Weigh Cities’ Tobacco Ban Rules
The court’s decision is expected to clarify the scope of municipal “home rule” powers in Ohio and could influence how local governments regulate a range of industries and products.
-

Philippine Health Groups Call for Science in Nicotine Control
The growth of unauthorized vape sales emerged as a key issue, with participants calling for stronger enforcement.
-

Dutch Report Raises Concerns Over Big Tobacco Getting into Cannabis
Researchers cited in the report raise concerns about industry-funded studies and communications shaping cannabis narratives.
-

Pentagon’s Nicotine Policy Called into Question
The article concludes that the DOD must better align clinical policy with operational reality to maintain credibility.
-

Harm Reduction Advocates Slam WHO’s First Nicotine Pouch Report
The World Health Organization’s first-ever report dedicated to nicotine pouches, a 152-page document titled “Exposing marketing tactics and strategies driving the global growth of nicotine pouches,” which was released May…
-
Minnesota Communities Push Flavored-Vape Bans as FDA Eases Restrictions
As the Trump administration signals a softer stance on flavored vapes , anti-tobacco advocates in Minnesota are intensifying efforts to restrict flavored nicotine products at the local level. A coalition…
Thought Leaders
-

A New Direction for ENDS
Chris Allen explains the significance of FDA’s shift on flavored vapes and why it reflects a growing emphasis on evidence-based harm reduction, behavioral science and product safeguards.
-

The Conglomerate Cigar Brands Are Losing Inside ChatGPT — and the Retailer Channel Is the Reason Why
Cigar Aficionado’s 2025 Cigar Insider Retailer Survey told the same story Cigar Insider has told every year for the last decade. Padrón at #1, Arturo Fuente at #2, Drew Estate…
-

The FDA’s evolving approach to youth-risk mitigation
Lilian Ortega explains the latest developments from regulators across pouches and ENDS and the latest enforcement of rules in the United States.
-
The Arithmetic of Harm
Why the World Cannot Afford America’s Regulatory Model By Dr. J. Preston Campbell, Cancer Researcher, Harm Reduction Scientist Heavy lifetime smoking kills approximately half of long-term users. That is not…
-

The Butt Stops Here
Because filters are small, lightweight, and routinely discarded in public spaces, they are disproportionately represented in stormwater runoff and shoreline debris counts despite their size.







